Read in
 English
English
Ik heb nog maar sinds een jaar een diagnose, en nu al staat er een grote medische doorbraak voor de deur: een kankermedicijn (momenteel nog in experimentele fase) toont verbazingwekkende resultaten bij overgroeisyndromen.
Dit nieuws werd vorige week donderdag bekend gemaakt, een bijzondere dag voor mij en al mijn lotgenoten die kampen met een ‘overgroeisyndroom’ veroorzaakt door het PIK3CA-gen. Dit gen regelt celgroei en veroorzaakt in ons geval overmatige groei in bepaalde delen van het lichaam.
In mijn geval gaat het om een goedaardige tumor in het gezicht (aandoening genaamd Facial Infiltrating Lipomatosis), maar er zijn ook mensen waarbij andere lichaamsdelen aangetast worden (aandoening genaamd CLOVES). Vaak kampen zij hierdoor met levensgevaarlijke problemen, van scoliose tot aan misvorming van de organen zoals de hersenen en nieren. Zij leven met chronische pijn en worden flink aangetast in hun dagelijks leven.
PIK3CA-gen in kanker
Het PIK3CA-gen komt ook voor bij bepaalde soorten kanker, zoals borstkanker en darmkanker. Daar wordt natuurlijk onderzoek naar gedaan. Zo werkt het Novartis laboratorium momenteel aan een inhibitor (remmer) voor het PIK3CA gen-in bovengenoemde kankersoorten. Dit medicijn (genaamd Alpelisib / BYL719) is momenteel nog in de experimentele fase.
Kankermedicijn werkt ook bij overgroeisyndromen
Eind 2015 behandelde een Franse specialist op het gebied van PIK3CA een 29-jarige patiënt met met een levensbedreigende vorm van CLOVES. Aangezien onze aandoeningen dezelfde PIK3CA-mutaties hebben als bovengenoemde kankersoorten, heeft de Franse dokter contact opgenomen met het laboratorium. In 2016 kreeg de dokter toestemming van het ANSM om het medicijn te testen op de doodzieke CLOVES-patiënt. Enorm goede resultaten volgden: de tumor was enorm afgenomen en het leven van de patiënt was aanzienlijk verbeterd. En ook heel bijzondere: na 18 maanden waren er geen bijwerkingen geconstateerd.
Proeven op muizen
Om meer inzicht te krijgen in de aandoeningen en de werking van het medicijn, creëerden de Franse dokter en zijn team een muismodel met alle symptomen die werden waargenomen bij de patiënt. Ook bij de muizen met CLOVES bracht het medicijn goede resultaten.
Toen er in 2016 weer een doodzieke 9-jarige patiënt aanklopte, kregen ze opnieuw toestemming om het experimentele medicijn toe te dienen. Ook nu bracht het medicijn spectaculaire resultaten zonder bijwerkingen.
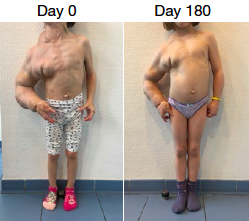
© Dr Canaud, AP-HP
Toestemming om het medicijn op meer patiënten te testen
Als gevolg van deze drie positieve resultaten, kregen ze toestemming van het ANSM om verder te testen op 17 patiënten met CLOVES. 14 kinderen en 3 volwassenen met levensbedreigende vormen van deze aandoening kregen het medicijn.
Bij allen van hen nam de tumor af, kregen de ledematen een normalere vorm en werden de problemen met scoliose minder. Ze konden zelfs stoppen met het gebruik van morfine en weer terug naar school. Operaties die gepland stonden, konden worden afgezegd.
Geen bijwerkingen
Na 18 maanden zijn er nog geen bijwerkingen geconstateerd. Wel hadden 3 van de 19 patiënten last van blaren in de mond en had 1 patiënt met obesitas last van een hoog bloedsuikergehalte, wat in bedwang wordt gehouden met een dieet. Tot nu toe moeten ze het medicijn de rest van hun leven slikken, anders groeit het weer terug.
Hoop voor de toekomst
We zijn er natuurlijk nog lang niet, maar het geeft wel enorm veel hoop voor de toekomst. Het medicijn moet eerst goed getest worden op bijwerkingen op de lange termijn. Daarnaast moeten patiënten het medicijn hun hele leven gebruiken. Het is nog even afwachten welk effect het medicijn gaat hebben op mijn lotgenoten met Facial Infiltrating Lipomatosis, aangezien het tot nu toe alleen bij CLOVES-patiënten gebruikt is.
Het is een enorme opluchting voor mijn CLOVES-lotgenoten die met de rug tegen de muur staan. Er is nu in ieder geval een middel dat hen kan helpen wanneer het nodig is. De resultaten van dit onderzoek geven hoop en ik ben heel benieuwd welke (betere) varianten er beschikbaar komen in de (hopelijk nabije) toekomst.
Lees meer:
> Een Frans artikel over deze doorbraak inclusief foto’s van voor en na de behandeling. Via chrome kan je de pagina automatisch laten vertalen.
> Engels artikel in Nature: “A medical first: CLOVES Syndrome and overgrowth syndromes: remarkable improvement in the health of 19 paediatric and adult patients using a new therapeutic strategy”

